Research Interests
- Development of multi-scale models and simulation of biological pathways and systems
- Use of simulation-based models of host-pathogen interactions to understand molecular mechanisms of pathogenesis and disease
- Development of integrated quantitative/empirical platforms to enable predictive modeling and simulation of host-pathogen and multicellular interactions by enabling acquisition of high-resolution kinetic, whole-cell data
- The use and application of information theory, coding theory, and signal processing to the analysis of genetic regulatory mechanisms
- Algorithm development for computational biosensors for detection and classification of polymorphisms, microbial identification and strain classification
Areas of Research
Multiscale Systems Biology and Immunopathogenesis
Host-Pathogen Coupled Networks
(Sponsor: Defense Threat Reduction Agency)
The goal of this work is the development of integrated models of host-pathogen networks critical in understanding the outcome of infection by various pathogenic agents, specifically the intracellular pathogens, Francisellatularensis(FT), Yersinia pestis(YP), and Burkholderia species. Our lab's contribution to this multiscale effort is in the development of pathogen specific response models, determination of interface models to integrate host response and bacterial adaptation, and computational tools and informatics platforms for model generation, implementation, and testing. This project is in collaboration with P. Robinson, J. Gearhart, and E. Hack (WPAFB/HJF, lead institution).
Systems Biology in 3D : Monitoring and Modeling Dynamics of Francisellatularensis-associated Granuloma Formation
(DOE/Sandia Lab Directed Research and Development)
The objective is to develop computational and experimental platforms that will enable the construction of theoretical models of the host response and granulomatous structures in Ft-infection. Specifically we are exploring mechanisms related to: 1) the functional role of macrophages and effector molecules during immune response; 2) the response of Ft to changing environments. This multidisciplinary project is in collaboration with several Sandia scientist and labs/groups and the University of New Mexico Health Science Center.
Analyzing the Genetic Basis of Mtb Latency and Reactivation
(NIH K25 Mentored Quantitative Research Career Development Award; Sponsor National Institutes of Health’s National Heart, Lung, and Blood Institute)
We are using BioXyce to simulate latent tuberculosis infection (LTBI) in a murine model of Mtb infection. BioXyce models of Mtb-host (the focus is on murine macrophages) interactions during latency will be used to generate empirically testable hypotheses regarding genetic and metabolic pathways in M. tuberculosis that contribute to LTBI and key signal transduction pathways in the mouse that impact the latency/reactivation process. NIH K25 grant in primary collaboration with University of New Mexico Health Science Center and secondary collaboration with Los Alamos National Laboratories.
Computational Platform for Multiscale Systems Biology: The BioXyce Systems Biology Simulation Platform
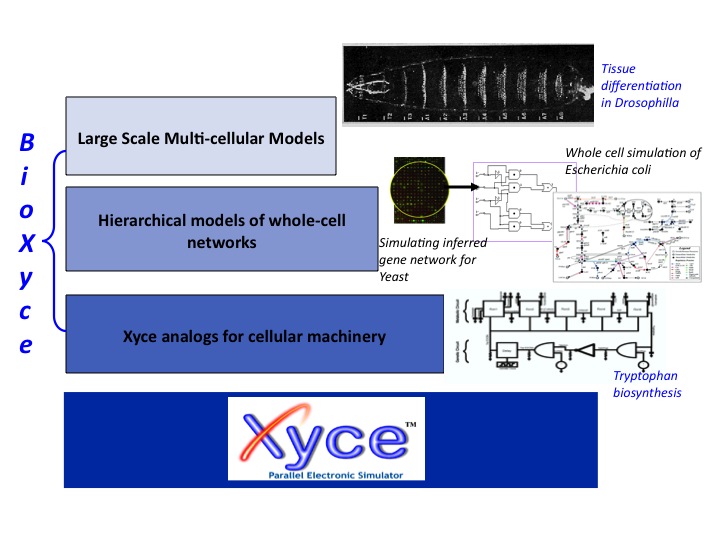 BioXyce is a biological network modeling tool that is based on the massively parallel modeling and simulation tool Xyce, used within Sandia and the Deparment of Energy (DOE) to perform massively parallel electrical circuit modeling [May and Schiek, 2009]. At the cellular level, biological regulation networks are modeled as electrical circuits where signals are produced, propagated and sensed. BioXyce uses the following equivalents: chemical mass as charge, mass flux as electric current, concentration as voltage, stoichiometric conservation as Kirchhoff’s voltage law, and mass conservation as Kirchhoff’s current law. With BioXyce, one can simulate large control networks consisting of entire cells, homogeneous cell cultures, or heterogenous interacting host-pathogen systems in order to understand the dynamics and stability of such systems. The input parameters for BioXyce, collected from literature and databases like BioCyc, KEGG, and BRENDA, are optimized using the DAKOTA (Design Analysis Kit for Optimization and Terascale Applications) UQ (uncertainty quantification) toolkit.
BioXyce is a biological network modeling tool that is based on the massively parallel modeling and simulation tool Xyce, used within Sandia and the Deparment of Energy (DOE) to perform massively parallel electrical circuit modeling [May and Schiek, 2009]. At the cellular level, biological regulation networks are modeled as electrical circuits where signals are produced, propagated and sensed. BioXyce uses the following equivalents: chemical mass as charge, mass flux as electric current, concentration as voltage, stoichiometric conservation as Kirchhoff’s voltage law, and mass conservation as Kirchhoff’s current law. With BioXyce, one can simulate large control networks consisting of entire cells, homogeneous cell cultures, or heterogenous interacting host-pathogen systems in order to understand the dynamics and stability of such systems. The input parameters for BioXyce, collected from literature and databases like BioCyc, KEGG, and BRENDA, are optimized using the DAKOTA (Design Analysis Kit for Optimization and Terascale Applications) UQ (uncertainty quantification) toolkit.
Systems Chemical Biology
Leveraging the BioXyce models of Mtb biochemical pathways involved in pathogen persistence, in collaboration with the Oprea Biocomputing Group (UNM-HSC) and the Tropsha Group (University of North Carolina – Chapel Hill) we are developing a systems chemical biology (SCB) platform to enable insight on the ability of small molecules to perturb and modulate microbial systems [Oprea et al., 2007; May et al. 2008; Faulon et al., 2008]. Systems chemical biology is a method for evaluating the effect of small molecules at the level of the entire biological system, by taking into account complex network relationships within the system.
Computational Genomics and Bioinformatic
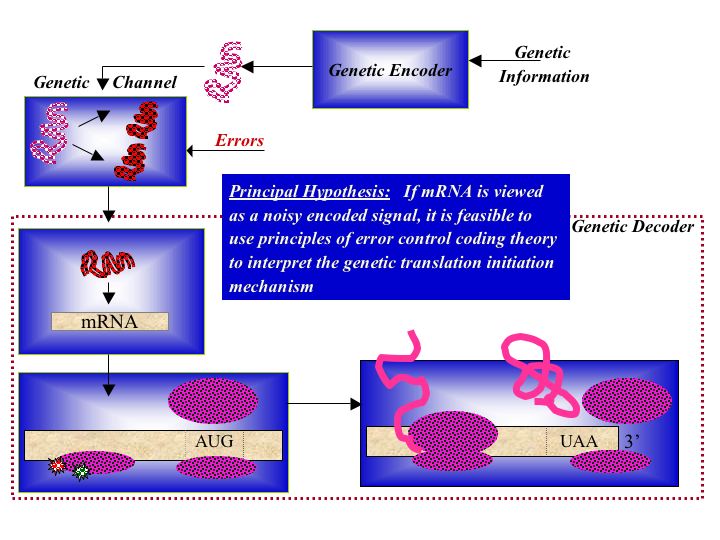 Biological Coding Theory
Biological Coding Theory
We are exploring the implications of understanding hybridization using the mathematical framework of error control coding theory and how this requires not only the use of information theoretic analysis tools, but compels us to view and model biomolecular systems as information transmission and processing systems. Using the genetic communication theory paradigm, we investigate coding theory algorithms for in silico categorization of single nucleotide polymorphisms based on the calculation of syndromes. We explore the use of coding theory frameworks in the design of in vitro computational biosensors, and design of error correcting biosensors for monitoring biomolecular systems.
Computational Biosensors
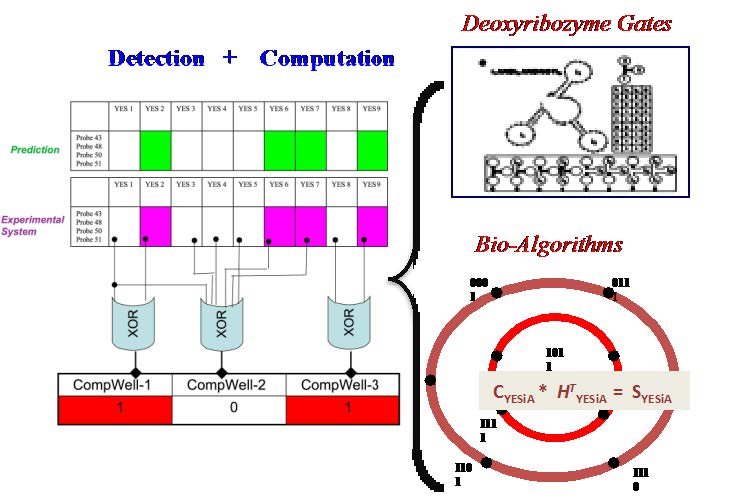 DNA-based Intelligent Microsensors for Genetically Modified Organisms
DNA-based Intelligent Microsensors for Genetically Modified Organisms
In collaboration with the Brozik Lab at Sandia National Laboratories, we performed research on the development of application-specific, rapid, stand-alone computational biosensors for concurrent detection and classification of nucleic-acid targets using deoxyribozyme molecular beacons [Stojanovic et al., 2001; May, Dolan, et al., 2008; May, Lee, et al., 2008]. Although single-stranded DNA sensor technologies, such as DNA microarrays, are widely used in transcriptome profiling and ultra high-throughput technologies are becoming more accessible, molecular beacon probes are highly sensitive and specific bioreceptors and enable the development of fieldablebiodetection systems [Wang 2000; Horejsh et al., 2005; Wu et al., 2007]. We have taken advantage of the deoxyribozyme molecular beacon’s computational capabilities and developed a biosensor system that can concurrently detect and, using deoxyribozyme logic gates,computationally classify targets in vitro [May, Lee, et al., 2008]. We have also demonstrated the ability to detect deoxyribozyme activity electrochemically, eliminating the need for fluorescence-based detection and reducing the noise in the empirical data [May et al., SNL SAND 2008].